 | |
| Clinical data | |
|---|---|
| Routes of administration | PO |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
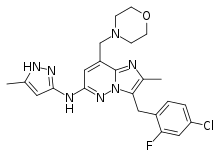
| Formula | C23H25ClFN7O |
| Molar mass | 469.95 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Gandotinib (LY-2784544) is an experimental drug developed by Eli Lilly for treatment of cancer. It is a small molecule JAK2 (Janus kinase) inhibitor, with additional minor inhibition of STAT3.
In a phase I trial, 16% of patients receiving the drug developed tumor lysis syndrome.[1] A phase II trial is underway for patients with myeloproliferative neoplasms, polycythemia vera, essential thrombocythemia, or myelofibrosis, who had failed ruxolitinib.[2][3]
References
- ↑ "Gandotinib - Eli Lilly". AdisInsight. Springer Nature Switzerland AG.
- ↑ Clinical trial number NCT01594723 for "A Study of LY2784544 in Participants With Myeloproliferative Neoplasms" at ClinicalTrials.gov
- ↑ Berdeja J, Palandri F, Baer MR, Quick D, Kiladjian JJ, Martinelli G, Verma A, Hamid O, Walgren R, Pitou C, Li PL, Gerds AT (August 2018). "Phase 2 study of gandotinib (LY2784544) in patients with myeloproliferative neoplasms". Leukemia Research. 71: 82–88. doi:10.1016/j.leukres.2018.06.014. PMID 30025280.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.